Document Type : Review Article
Authors
1 Center for Incubation, Innovation, Research and Consultancy, Department of Chemistry, Jyothy Institute of Technology, Thataguni, Bengaluru-560082, India
2 Visvesvaraya Technological University, Jnana Sangama, Belagavi, Karnataka 590018, India
Abstract
Animal fats have the potential to be a valuable resource in various fields due to their abundance and renewability. This review is aimed to highlight the potential of animal fats for industrial applications. Despite their versatility, the high levels of unsaturated fatty acids and instability have limited their usage. Chemical modification processes such as epoxidation, esterification, and acetylation are used to enhance stability and expand their applicability in various fields. These modified animal fats offer a sustainable alternative to petroleum-based products with a positive impact on the environment. The usage of animal fats in other industries provides more benefits, including reduced dependence on fossil fuels, savings in foreign currency, and an improved rural economy with increased job opportunities. In addition, the utilization of animal fats in the chemical industry can lead to the development of biodegradable products, which have a positive impact on the environment.
Graphical Abstract
Keywords
Main Subjects
- Introduction
usiness Wire, a Berkshire Hathaway Company reported on the topic “Animal and Marine Fats and Oils Market by Product Type, Source, Form and Application: Global Opportunity Analysis and Industry Forecast, 2021–2030”.
In 2020, the market size for animal and marine fats and oils was recorded as $222,335.0 million, and it is projected to grow to $516,759.3 million by 2030, representing a CAGR of 7.6% from 2021 to 2030. Due to the increase in animal fat production, it is concerning that the amount of waste from animal fats is also increasing, which can lead to environmental pollution [1]. The use of waste animal fats in the production of various industries other than food industries can suppress the food versus product competition, which can contribute to reduce food prices and avoid food insecurity for vulnerable populations [2]. For example, animal fat, extracted and purified through the Coctio bone broth process, serves as a by-product derived from animal bones. It has versatile applications and can be supplied to the biodiesel industry as a non-edible component for fuel production. In addition, the refined animal fat finds utility in the food, cosmetics, and pharmaceutical sectors [3]. This article highlights the underutilized potential of animal fats, which are considered waste oils, in various industries such as food processing industries (vegetable oil refining residues, spent frying oils, and oil rich by-products) [4], agro-industries (agricultural crops such as oil seeds which generates oil-rich byproducts) [5], animal rendering industries (fat trimming and used cooling oil) [6], and pharmaceutical and cosmetic industries (residues from oil extracts) [7]. The review sheds light on the opportunities for future advancement and creativity in diverse domains, utilizing the results to discover new and innovative applications for raw materials that are considered waste. The article emphasizes the importance of interdisciplinary research in shaping various aspects of society and highlights the significance of animal fat sources, classifications, production cycles, chemistry, potential chemical reactions with fats, available fatty acids, and industrial applications in bringing out eco-friendly products.
Finally, industrial applications of animal fats are of great interest to bring out eco-friendly products which are reviewed with effort. The graphical abstract shown the production, reactions and their applications of the animal fats.
- Classifying Oils- An Insight into Different Oil Varieties
Oils are mainly extracted from seeds (plants) and fats (animal) sources. Based on their origin, composition, and intended use, oils are categorized into edible, non-edible, and animal fats [8]. (1) Edible oils, extracted from different plant-based sources, such as fruits, seeds, and nuts, contain essential fatty acids and provide many health benefits. Edible oils are suitable for human consumption. Examples of edible oils include canola oil, coconut oil, vegetable oil, and peanut oil. (2) Non-edible oils from plant sources are not suitable for human consumption as they have a bitter taste, a pungent smell, and are toxic. Examples of non-edible oils include jatropha, castor, and pongamia. (3) Animal fats are derived from animal sources such as tallow, beef, lard, pork, butter, and poultry and are used in baking and cooking.
It is significant to mention that some fats and oils can belong to multiple categories based on their specific application. For instance, lard can be used both as an edible fat for cooking and as a non-edible oil in the manufacture of soap and other products.
- Livestock into Fat- An Enlightenment into the Animal Fat Production Cycle
The animal fat production process is a complex and multi-step process that is thoroughly explained in the below subsections [8].
3.1. Production of milk fats
Cream and non-fat milk layers are separated from milk using a centrifuge or gravity-based separator, and then cream is churned by hand or using a machine until it becomes solid. This creates friction through high agitation of the cream, which leads to the fat to separating from the buttermilk. The obtained melted fat is called butter fat [9-10].
3.2. Production of rendered oils
The process of rendering animal fats into greaves involves several key phases. Adipose tissues are subjected to the disintegration (shredding, grinding, or milling) process to break down the tissues into smaller pieces, making it easier to extract the oil. These tissues are allowed to melt to release the oil. This process involves heating the tissues to a high temperature, which separates the fat from the solid materials by melting. Finally, centrifugation or filtration is to separate oil from the solid materials. Separated oil (tallow or lard) is then subjected to a clarification (de-waxing, de-gumming, and de-acidification) process to improve its quality. The leftover solid materials obtained are called Greaves meal. This byproduct can be used in animal feed as a protein source [11].
3.3. Production of fish oils
The production of fish oil from fish typically involves the following steps. The initial stage is to soften the flesh by cooking and remove any unwanted impurities or bacteria. This is often done by steaming or boiling the fish. The next stage is to separate the oil from the fish flesh. This is typically done by pressing the fish flesh to extract the oil or using solvents to dissolve the oil. Once the oil has been separated, it is usually subjected to a clarification process to remove any remaining impurities. This may involve filtering the oil through a series of filters or using centrifugation to separate the oil from any solid particles. The clarified oil is then ready for further processing, such as refinement and bottling [12].
- Chemistry behind Animal Fats and Oils
The presence of fatty acids within fats and oils triggers various types of reactions. The most common reactions are hydrogenation, ozonolysis, transesterification, epoxidation, oxidation, hydrolysis, and saponification [13]. The detailed chemical reactions are demonstrated in Figures 1-6. Hydrogenation involves the transfer of hydrogen atoms from the H2 gas to the double bonds in the unsaturated fatty acids. This leads to the formation of single bonds, decreases the number of double bonds, and increases the saturation degree of the fatty acids. The result is a change in the physical and chemical properties of the fatty acids, which converts their phase from liquid oils into solid fats [14]. These hydrogenated fats are used in processed foods to increase the life span and also used instead of frying oil to withstand the heat and can be used longer [15]. Hydrogenated fats contribute to the texture and mouthfeel of processed foods. Trans fats produced during hydrogenation have been linked to increased cardiovascular risks [16]. As an alternative, it is used for the production of precursors in the production of biodiesel and bioplastics in recent years [17].
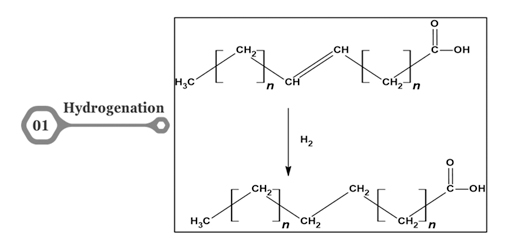
Figure 1. Hydrogenation
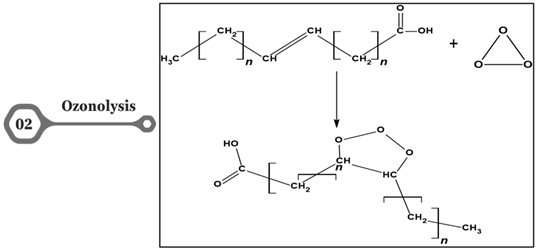
Figure 2. Ozonolysis
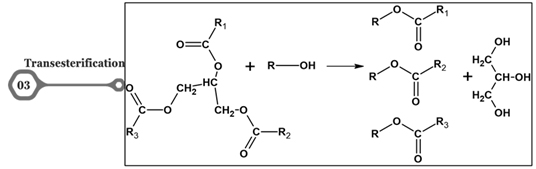
Figure 3. Transesterification

Figure 4. Epoxidation
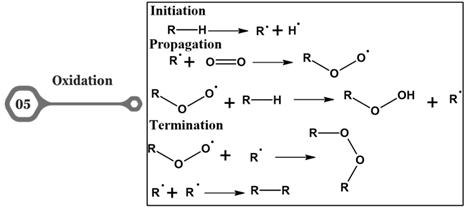
Figure 5. Oxidation

Figure 6. Hydrolysis/Saponification
Ozonolysis is a chemical process that involves the reaction of ozone (O3) with unsaturated compounds. This process is utilized for various industrial applications, leading to modifications in the chemical structure of compounds and generating value-added products and intermediates. The modifications resulting from ozonolysis have specific effects on the performance of fats, oils, or other compounds, and these effects can vary based on the nature of the starting material. Compared to many oxidation routes, ozone is produced on site eliminating problems related to storage and transportation of oxidation agents [18].
Transesterification is a process where the animal fats (triglycerides composed of fatty acids and glycerol) react with an alcohol (methanol or ethanol) to give fatty acid methyl or ethyl esters (biodiesel). This process is used to produce biodiesel from different feedstocks. The primary effect is the conversion of animal fats into biodiesel, a more environmentally friendly and sustainable fuel alternative. Biodiesel has lower viscosity compared to animal fats, improving its flow characteristics and ease of use [19].
The addition of an oxygen atom to the carbon-carbon double bond present in the fatty acid using reagents such as peroxy acids or peracids to form epoxide group is known as epoxidation. This reaction will modify the physical properties of the oil and fats and improve the oxidative stability. This modified oil and fats can be used in the industries for the production of plasticizers, resins, and lubricants [20]. The oxidation of fatty acids in fats and oils takes place when they are exposed to air, light, or heat. They form a variety of by-products like aldehydes, carboxylic acids, peroxides, and ketones. These by-products have a negative impact on the quality or stability of the oil and leads to the formation of off-flavors, and rancidity. This can be minimized by treating it with antioxidants and recently encapsulation technologies [21]. Hydrolysis/Saponification occurs when fats or oils are exposed to water and base, the ester bonds between the fatty acids and glycerol are broken down. This reaction can result in the formation of sodium salts of the particular fatty acids (soap) and glycerol. The commercial application in this process is the production of soap [22].
4.1. Fatty acids in animal fats
Animal fats consist of lipids with a variety of fatty acids. Fatty acids serve as the foundational elements that differ in their saturation, structure, and length. Understanding the constituents of fatty acids is significant and provides a way to different applications namely caproic acid is one of the components in the vanilla and cheese and also used as an artificial flavor when it is changed into its ester form which can be used in food industries [23]. Caprylic acid is the saturated one which has the antimicrobial properties which is used as an antimicrobial agent in pharmaceutical industries [24]. Lauric acid which is used as a good antibacterial agent because it destroys the cell wall and the membrane of the bacteria [25]. Using more of myristic acid in the diet, immunomodulatory functions are exerted [26]. Palmitic acid has been used as the milk replacers for the young feeding animals. Palmitic and stearic acid has been used in the lactating cows to increase the nutritive value in its diet and enhance its milk production [27]. Oleic acid is mostly present in the animal fats which is used to treat the coronary heart diseases. Linolenic acid is used as a supplementary diet for the animals which itself cannot synthesize this fatty acid [28]. Types of fatty acids found in animal fats including information on their source, chemical name, molecular weight, chemical formula, number of double bonds, and structure are summarized from suitable research articles, as presented in Table 1 [29-37].
Table 1. Composition of animal fats: A comprehensive study of fatty acids and their characteristics [29-37]
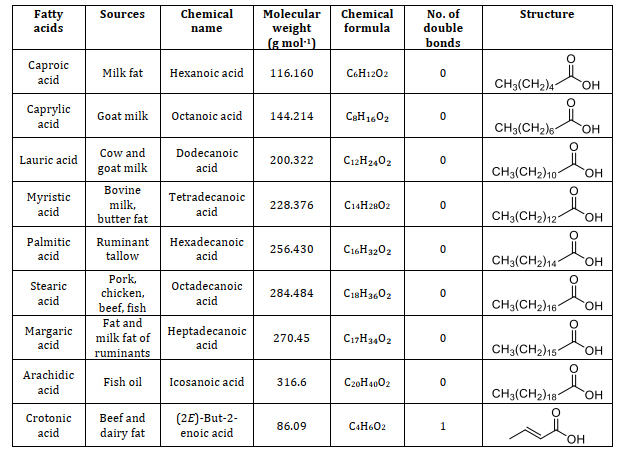
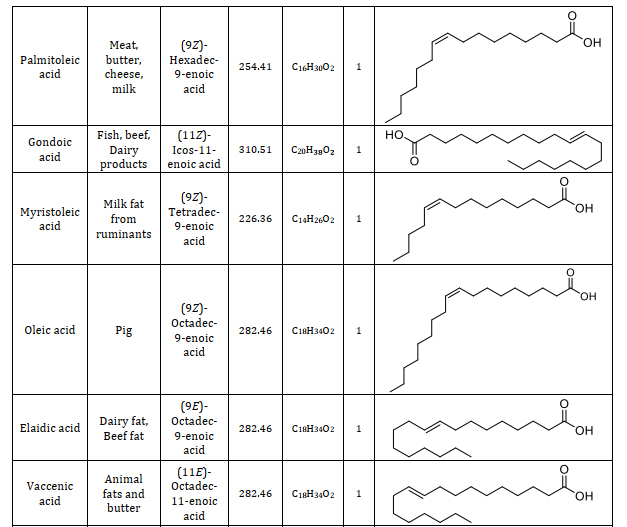
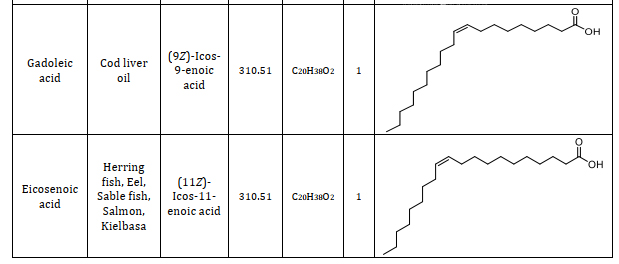
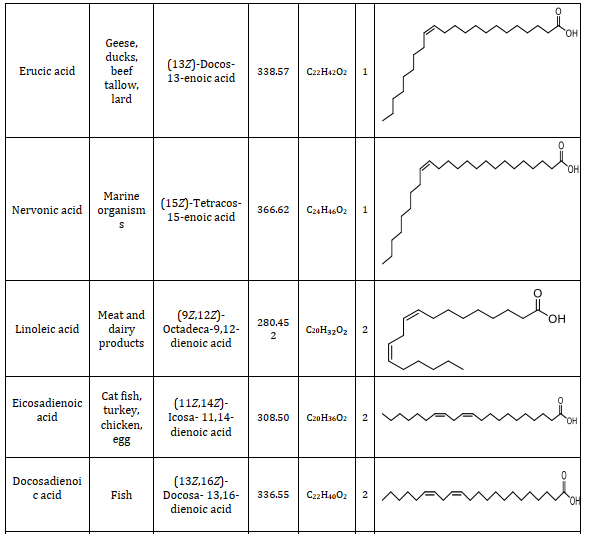

4.2. Fatty acid profile analysis of animal fats
Animal fats contain a spectrum of fatty acids, including saturated, monounsaturated, and polyunsaturated types. Understanding the fatty acid composition in animal fats offers diverse practical applications across various domains. Ternary plot in Figure 7 illustrates the fatty acid composition of different animal fats [38]. The composition of fatty acids varies among animal fats, influencing their applications. The ternary plot provides a visual representation enabling a quick assessment of the overall fatty acid content.

Figure 7. Ternary plot of the number of different types of fatty acids in animal fat
- Applications of Animal Fats
The modified animal fats produced from the various chemical process mentioned in Section 3 are more stable than the ordinary animal fats which can be used in various applications. The detailed explanation with recent literature survey has been discussed in the upcoming subsections.
5.1. Lubricant
Lubricants are crucial in all mechanical systems and reduce the wear and friction of moving parts between metal surfaces. Common industrial lubricants include metalworking fluids, engine oils, gearbox oils, compressor oils, and hydraulic oils. Nearly 50% of petroleum lubricants cause permanent damage to the environment. This has led to increased demand for biodegradable and renewable lubricant sources. A variety of oils are converted into biodegradable lubricants using methanol [39-40]. The process of producing biolubricants involves various reaction pathways, as shown in Figure 8. Properties such as friction and wear, oxidation stability, biodegradability, and environmental compatibility are important for effective performance as a lubricant. Animal fats have been found to have the potential to be used as biolubricants discussed in the next paragraph [41].
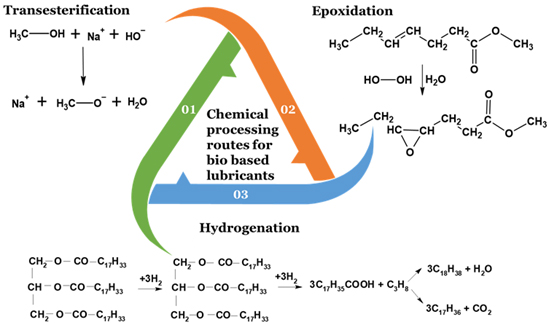
Figure 8. Reaction routes for the preparation of biolubricants
Lubricant from the chicken fat was obtained by physical refining process. 30-40% chicken fat was obtained from the poultry waste and converted into esters by the transesterification process. The esters viscosity is varied using oxidative stabilizers and viscosity modifiers such as ethyl vinyl acetate (EVA) and styrene butadiene styrene (SBS). The tribological properties of the samples (pure biodiesel, pure chicken fat, and various blends of biodiesel plus chicken fat with different compositions of SBS and EVA were compared [42]. Omega-3 fatty acids were separated from the waste fish oil and used in many nutrition products. The remaining fatty acids are considered waste and used as lubricants, biofuel, and many other applications [43]. Fish oil obtained from Nile Tilapia is chemically modified by esterification, epoxidation, and ring opening reactions. This modified fish oil is compared with commercial lubricants and it showed high biodegradability and viscosity [44]. Duck fat biodiesel used as a biolubricant and compared with other vegetable oils such as, soybean, olive, coconut, palm, and canola [45].
5.2. Fuel
In response to the growing demand for oil, the Indian government [46] has passed an order to promote the mixing of higher levels of ethanol and vegetable oil components with diesel and gasoline. Ethanol (10%) blended with gasoline has increased from 12% to 15%. For diesel, 20% of alkyl esters are from long-chain fatty acids of oils taken in the upcoming years. This move is aimed at reducing the country’s dependence on fossil fuels and promoting eco-friendly alternatives [47] and strategies to blend is explained in this article [48]. Rudolph Diesel was the first person to use oils in diesel engines in 1911. The properties of raw oil are same as crude petroleum derived diesel and used in diesel engines without any blending. However, using the oils led to several drawbacks such as more carbon deposits, oil ring sticking, injector coking, high fuel viscosity, etc. Few of them can be solved, like reducing the viscosity by various methods (micro-emulsification, pyrolysis, dilution, and transesterification). Among these methods, transesterification is considered the best [49]. The biodiesel production by the transesterification process is elaborately explained in Figure 9 [50-51]. The research work from was 2020 to 2023 for the biofuel production from different animal fats were listed in Figure 10 [52-66]. It is evident from figure that utilization of animal fat acts as a key source for biodiesel production.

Figure 9. Transesterification process for biodiesel production

Figure 10. Review on animal fat sources for biodiesel synthesis from 2020-2023
5.3. Plasticizer in films
Plastics have entered in different ways into human lifestyle when standard of living gets modernized. But concerning the environmental account, it causes global pollution due to its non-biodegradable nature. If plastics usage goes on without limit, it will lead our world into a “plastic Earth” [67]. Therefore, there is a need to transition to biodegradable products. Biodegradable starch has been widely used in packaging industry due to its low cost and abundance in nature, but it has poor hydrophilicity and low extensibility as its shortcomings. Other than starch, many biodegradable polymers have been used, such as chitosan [68], polylactic acid, polycaprolactone, etc. To meet the issues, plasticizers came into action. The plasticizers used were glycerol, esters, and sorbitol. Among these plasticizers, glycerol is toxic-free and cost-effective. The main criterion for the film to get onto the market is that it should be water resistant. To make the film waterproof, oils have been used as an additive.
Several investigations have utilized plant oils in film preparation, including soybean oil [69], bamboo volatile oil [67], palm oil [70], cinnamon oil [71], olive oil [68], and buriti oil [72]. These studies employed the solution casting technique for the preparation. The basic steps involved in the formation of film by solution casting technique are pictorially demonstrated in Figure 11. Most of the films were done with the help of vegetable oil, but only few animal fats namely shellac resins from Laccifer lacca used as a coating on the surfaces of tomatoes [73] and green chilies [74].
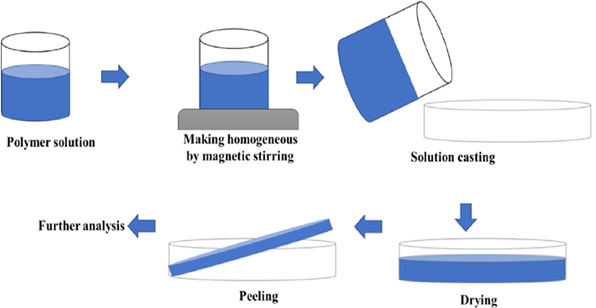
Figure 11. Schematic representation of solution casting technique for film preparatio
5.4. Cutting fluid
The cutting process results in getting the desired shape and geometry of the specimen by using sharp machining tools. The energy required for such an operation is almost 66%, and major energy loss happens in the form of heat. The heat loss can be minimized by the use of metal cutting fluids (MCF) [75]. The MCF working is elucidated in Figure 12 [76]. Mineral oils were used as MCF which severely reflects on the machine operator and the environment. The people and the environment exposed to the non-biodegradable metal cutting fluids suffer from cancer, lung and heart disease, DNA damage, skin irritation, and soil contamination due to disposal. The role of cutting fluid is to increase the life span of the tool by sluicing the chips from the cutting zone, reducing the thermal deformation of the workpiece, and improving the surface finish [77].

Figure 12. Working of metal cutting fluid (MCF)
The reason for using mostly vegetable oils as metal cutting fluids is because that they stay in liquid form. The properties of the adsorption of oil facilitate the adsorption on the surface of the friction. This property provides a protective layer when undergoing machining operations. Not only is it good for lubrication, but it also has the properties of biodegradability and non- toxicity [78]. Animal fats modified by undergoing rendering, steaming, and extracting the oil, which is then used for the emulsification process. Tallow and lard were explored as potential MCF [79].
5.5. Wound dressing
Infections delay the progress of wound healing and even cause sepsis. Antibacterial wound dressings are made to reduce or kill microbes during the healing time. The dressings were made to be biocompatible, provide moisture over the wounded tissues and protect the new tissues formed during the healing process from external trauma. To lessen the usage of fossil-based raw materials for polyurethane synthesis, bio-based feedstocks like plant oils were used [80-81]. Animal fats such as emu oil and badger oil themselves cannot be used as wound dressing components, so to provide support to the composites; they are blended with polyurethanes [82-83].
In addition, nanoparticles which have antimicrobial properties were loaded along with the composites [84]. Those mixtures were taken in the syringe to undergo the electrospinning process as shown in Figure 13 to form wound dressing material. Various essential oils were evaluated their antimicrobial properties for wound dressing application [85-86].
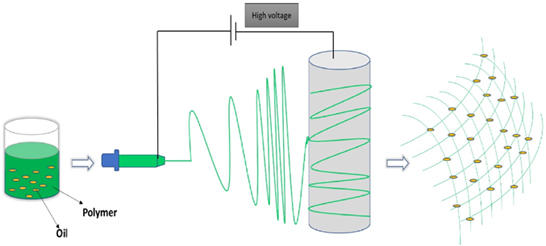
Figure 13. Pictorial representation of the electrospun process of wound dressing
5.6. Paints
In ancient times, the practice of using oils as a binder for pigments was followed. The Egyptians around 2600 BCE used a mixture of oil and pigments to create a creamy paste for inlay work [87]. Linseed oil was the most widely used medium for binding the paints in Europe, while poppy and walnut oil was more expensive options. Fish and marine mammals contain ample amounts of oil to utilize by coastal communities as paint binders or varnishes. However, whale oil has good commercial value, so it was used in a limited amount [88-89]. In the US, fish oils, particularly from menhaden (Brevoortia spp.), became heavily exploited due to the abundance availability of this silvery fish along the Eastern and Atlantic seaboard [90]. Despite being considered unappetizing for human consumption, menhaden were utilized as bait or processed for their oil and used in various products such as margarine, soap, and paint [91]. The free fatty acid percentage profile of the fish menhaden is represented in Figure 14 [92].
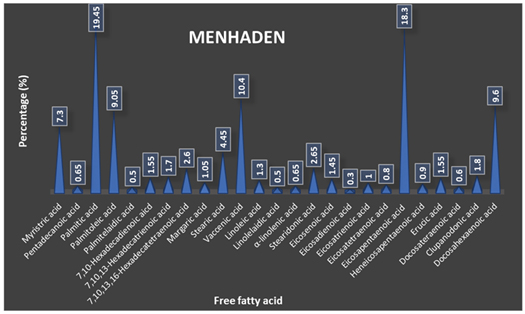
Figure 14. Free fatty acid percentage profile of menhaden (Brevoortia sp.)
5.7. Cosmetics
In the cosmetic industry, rendered poultry fat, tallow, and lard are widely used. Lard is obtained from the rendering of swine adipose tissue, while tallow is obtained from the rendering of ruminant fat from farm animals such as sheep, buffalo, cattle, and goats. These rendered fats possess occlusive, emulsion-stabilizing, emollient, surfactant, and viscosity-increasing properties. Modern cosmetic products utilize fat from various animal species are illustrated in Figure 15 [93].

Figure 15. Applications of various animal fats in cosmetic industry
Animal fats such as emu oil and mink oil have penetrating power and easily get inside the dermal layers, stimulating hair and skin growth. These can be used in making lipsticks, hair sprays, moisturizers, body lotions, and skin cleansers [94]. Ostrich oil is used in antiaging cosmetics because it is rich in oleic acid, lauric acid, and palmitic acid [95]. Horse oil has skin moisturizing and skin barrier restoration properties and is used in making soap, shampoo, lotion, and face cream [96].
Ambergris wax is a constituent of perfumes that prolongs their scent. Sperm oil is used in beauty products as a moisturizer and as a vehicle for fragrances. Cod liver oil is used as an occlusive in skin care products. Shark liver oil is a triterpenoid organic compound that impressively mimics the skin’s natural oils and is used in lip balm, hair dyes, facial moisturizers, sunscreen, and moisturizing creams. Crocodile oil is rich in oleic acid and Ω-3, 6 fatty acids. Due to its good healing property, it is used in skin care products [97].
- Conclusion
Based on the overview elucidated, the animal fats pave a way for the search of inventing new products. This paper mainly focuses on ecofriendly and biodegradable things discovered recently. Edible oils that are suitable for consumption are not favored due to their higher cost. However, nonedible animal fats have been identified as potential sources for various applications. Animal fats have proven to be a versatile and indispensable resource in automobiles (lubricants, additives in fuels, and components in car care products), cosmetics (production of soaps, creams, lotions, and other beauty and skincare products), food (production of margarine, shortening, cooking oils, and flavorings), paints (binders, solvents, or additives, providing improved performance and durability), and pharmaceutical industries (pharmaceutical ointments, capsules, and coatings for various medicinal products). By addressing these specific points, animal fats can be recognized as valuable and underutilized resources across industries, promoting their responsible usage and maximizing their economic and practical benefits.
Acknowledgements
The authors acknowledge the support from Centre for Incubation, Innovation, Research and Consultancy for successful completion.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Orcid:
Nandhini Dhanavel: https://orcid.org/0009-0001-4128-4287
Meenakshi Halada Nandakrishnan: https://orcid.org/0000-0002-4010-9495
Citation: N. Dhanavel, M.H. Nandakrishnan, A Review of Animal Fat: A Great Source for Industrial Applications. J. Chem. Rev., 2024, 6(2), 115-137.
----------------------------------------------------------------------------------------------------------------------------------------------------
OPEN ACCESS
©2024 The author(s). This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit: http://creativecommons.org/licenses/by/4.0/
PUBLISHER NOTE
Sami Publishing Company remains neutral concerning jurisdictional claims in published maps and institutional affiliations.
CURRENT PUBLISHER
Sami Publishing Company


